The Golden Age of Biobanking: A Double-Edged Sword
We are in a pivotal era of medical innovation, where biobanking has become the cornerstone of biomedical science. From precision medicine to AI-driven drug discovery and advanced therapeutics, biobanks now serve as the foundational infrastructure of personalized healthcare.
Modern biobanks are complex systems that manage the full lifecycle of biospecimens, from collection and processing to storage and global distribution. These biological samples, such as blood or tissue, are linked with clinical data, generating massive volumes of highly sensitive information. Institutions like the UK Biobank and NIH’s All of Us program now manage petabytes of such data using robotics and advanced informatics.
The emergence of virtual and networked biobanks, connecting institutions across borders, has supercharged data sharing for research into rare diseases and large-scale genomics. But the very attributes that make biobanks powerful —scale, interconnectedness, and data intensity, also expose critical vulnerabilities.
The legacy systems designed for standalone repositories are failing under the weight of modern requirements. The result is a trust crisis, not in science itself, but in the foundational infrastructure underpinning it. The industry’s most valuable asset, its data, has become its greatest liability.
What is the Trust Crisis in Biobanking?
The rapid evolution of biobanking has surfaced three systemic, interlocking challenges:
The Compliance Crisis: Navigating a Global Legal Minefield
Regulations are no longer simple checklists; they are a complex, overlapping web of global mandates. While frameworks like the EU’s General Data Protection Regulation (GDPR) and the US’s Health Insurance Portability and Accountability Act (HIPAA) command attention, the reality is far more granular. Biobanks must also contend with the FDA’s 21 CFR Part 11, which dictates the trustworthiness of all electronic records and signatures, and the ISO 20387 standard for biobank quality and competence. Adding to this complexity, nations like India have enacted their own robust legislation, such as the Digital Personal Data Protection Act (DPDPA), 2023.
These standards were never designed for today’s networked, AI-driven research ecosystems, creating a minefield where demonstrating auditable proof of compliance is a constant struggle.
Impact: Organizations face substantial regulatory risk, including multi-million-dollar penalties, operational shutdowns, and reputational damage. For biotech firms, non-compliance jeopardizes access to critical datasets, delaying innovation and product development timelines.
The Patient Consent Crisis: Erosion of the Donor Compact
The growing risk of genomic re-identification has rendered traditional one-time, blanket consent models obsolete. Today’s donors expect dynamic, granular control over how their data and biospecimens are used. Yet, most legacy systems lack the interoperability required to manage and enforce these evolving consent directives across multiple systems and collaborators.
Impact: Any breach of donor privacy can erode public trust, drive participant attrition, and hinder recruitment for future studies. A biobank that fails to maintain this trust jeopardizes not only its relevance, but its very existence.
The Data Integrity Crisis: The “Garbage In, Gospel Out” Risk of AI
The potential of AI in biotechnology is immense, but its value is entirely dependent on the quality and integrity of its training data. This goes beyond just “messy data.” It’s about the undocumented freeze-thaw cycles that compromise sample viability, the disorganized freezer management that leads to lost aliquots, and the failure to capture standardized data points required by guidelines like BRISQ (Biospecimen Reporting for Improved Study Quality).
In regulated environments, these gaps break the fundamental principles of data integrity ALCOA+ (Attributable, Legible, Contemporaneous, Original, Accurate, plus Complete, Consistent, Enduring, and Available). Without a verifiable data trail, AI models risk being trained on flawed, non-compliant inputs.
Impact: The consequences include billions in wasted R&D, unreliable scientific outputs, retracted publications, and AI tools that cannot meet regulatory scrutiny. An product built on low-integrity data isn’t just uncertain, it’s a liability.

How Blockchain Solves Biobank Data Integrity Issues
Solving these crises requires more than incremental fixes. It demands architectural change. Blockchain, especially via Blockchain-as-a-Service (BaaS), delivers a foundation built on verifiable trust.
Key Benefits of Blockchain in Biobanking
- Immutable Compliance: Create tamper-proof, timestamped audit trails that log every sample transfer, consent update, and access event. This makes compliance with 21 CFR Part 11 and ISO 20387 verifiable, not just stated.
- Dynamic Consent and Decentralized Identity: Use smart contracts and Decentralized Identifiers (DIDs) to let donors manage digital consent in real time, increasing transparency and trust.
- Verifiable Data Provenance for AI Governance: Establish a complete, unbroken chain of custody for biospecimens and their associated data, from collection through every freeze-thaw cycle to AI training, enabling regulatory-grade transparency and adherence to BRISQ standards.
Real-World Examples of Blockchain in Biobanking
Blockchain is already in action. At a National Cancer Institute–affiliated breast cancer biobank, a decentralized mobile app using “biowallet” NFTs enabled more than 400 participants to track usage of over 1,800 specimens demonstrating scalable, real-world dynamic consent.
Additionally, Malta’s Dwarna portal securely records consent changes on-chain, providing donors with transparency and the ability to withdraw consent meeting GDPR’s erasure standards.
Blockchain also underpins explainable AI (XAI) initiatives. By logging every step in data processing and model development, it ensures auditability, reproducibility, and regulatory trust.
Blockchain-as-a-Service (BaaS): The Smart Path to Adoption
Building blockchain infrastructure in-house is prohibitively expensive for most biobanks. BaaS provides an efficient alternative, similar to how cloud computing revolutionized software infrastructure.
Benefits of BaaS for Biobanks
- Faster Deployment: Integrate in weeks, not months
- Lower Overhead: No major infrastructure investment needed
- Scalability: Start small and expand easily
- API-Driven: Connect seamlessly with your LIMS, EDC, and ERP systems
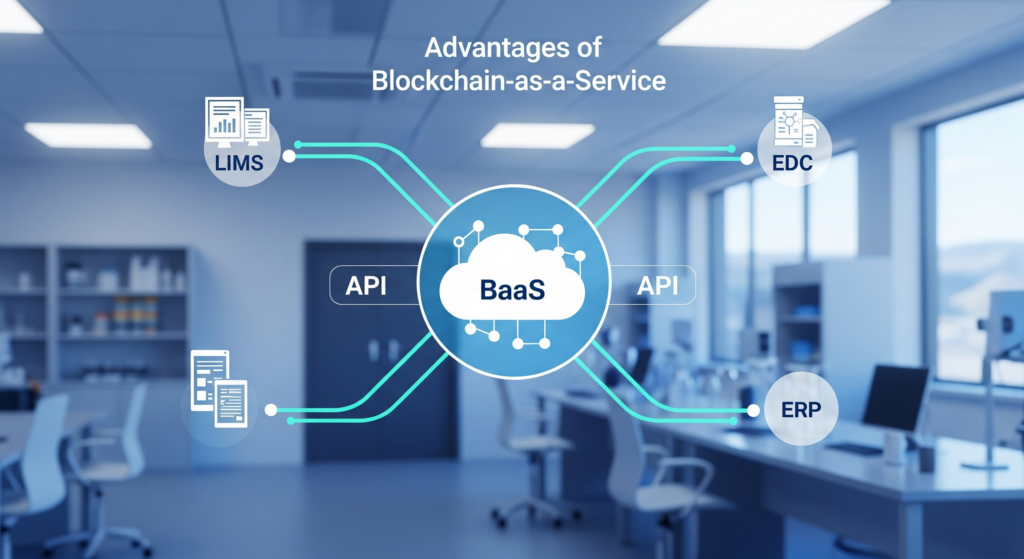
Operationalizing Blockchain: How FLEXBLOK Enables Biobank-Grade Adoption
Adopting blockchain in life sciences comes with real complexity: regulatory constraints, validation needs, and IT compatibility. FLEXBLOK was built to bridge that gap.
Rather than overhauling existing infrastructure, FLEXBLOK integrates with your existing systems like LIMS, EDC, and ERP via developer-ready APIs. This enables:
- Automated compliance tracking for regulated workflows, streamlining the audit process by providing a single, immutable source of truth.
- Dynamic consent deployment using decentralized identity standards that scale with large participant pools
- On-chain data provenance that supports 99.9 percent traceability and enhances AI model auditability for regulatory readiness
By focusing on operational compatibility and regulatory alignment, FLEXBLOK helps organizations build verifiable trust into every layer of their data pipeline without disruption.
Conclusion: From Promises to Proof
The era of data-intensive, AI-driven biomedicine demands more than trust—it demands verifiable proof. Biobanks must prove that their data, systems, and governance models meet the highest standards.
Those that embrace blockchain-powered infrastructure, especially through strategic BaaS platforms, will not only mitigate risk but lead the next generation of biomedical discovery.
Take control of your compliance, donor trust, and data integrity. Explore how FLEXBLOK helps you operationalize proof and confidently scale your biobank. Request your personalized demo now.


